Fourth state of matter: between solid and liquid
What is a liquid crystal?
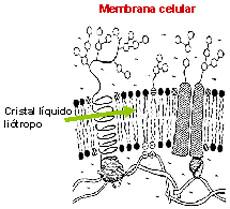
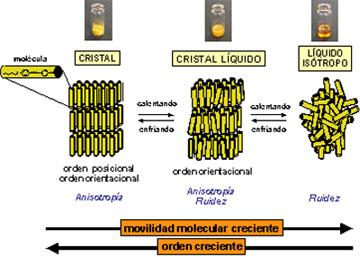
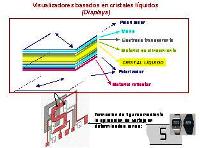
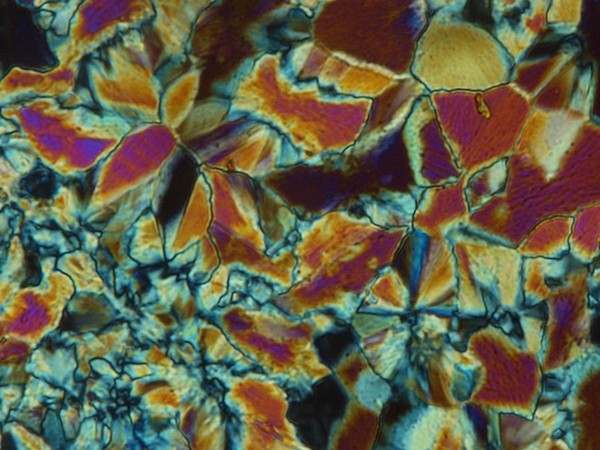
When you think about it, it is surprising to hear someone talk about liquid crystals. It is not easy to put the two words together when you consider what each of them means. However, although their knowledge is still somewhat limited, it is known that they owe their properties to the characteristic of some chemical compounds to present this fourth state of aggregation of matter.